PREVALENCE OF THYROID DYSFUNCTION IN PATIENTS
WITH RHEUMATOID ARHTRITIS
Tran Nguyen Quynh Tram , Nguyen Thi Bich Dao*
ABSTRACT
Introduction:The relationship between thyroid disease and rheumatic disorders has been the subject of considerable debate. Abnormal thyroid function and/or autoimmune thyroid disease were observed in 8% to 35% patients with rheumatoid arthritis (RA).Objectives: To evaluate the prevalence of thyroid dysfunctions in RA.Patients and Methods:This is a cross sectional analytical study. Thyroid hormones, anti-TPO as well as medical and RA-related history were evaluated in 113 RA patients.Results: 18 out of 113 RA patients (15,9%) had thyroid dysfunction. The most common thyroid dysfunction was hyperthyroidism (83,3%). Most patients with thyroid dysfunction were over 40-years-old,> 70% of them had moderate to high disease activity score (DAS-28) (over 70%), and 40% with goiter. Anti microsomal antibody was positive in 27 patients (23,9%) but only 9 patients (8%) were diagnosed with autoimmune thyroid disease. (AITD). Conclusions: Thyroid dysfunction commonly occurs in patients with RA. It is reasonable to include screening for the coexistence of thyroid disease(s) inthese patients in order to prevent the complications associated with thyroid dysfunction and to avoid adding to the morbidity of existing autoimmune rheumatic disease.
Keywords:autoimmune thyroid disease, thyroid dysfunction, rheumatic arthritis.
Main correspondence:Tran Nguyen Quynh Tram
Submission date: 8 Feb 2017
Revised date: 20 Feb 2017
Acceptance date: 15Mar 2017
I. INTRODUCTION:
Rheumatoid arthritis (RA) is a chronic autoimmune systemic inflammatory multisystem disease of unknown cause that may affect many tissues and organs, but principally attacks synovial joints, primarily affecting the peripheral joints in symmetrical pattern[9].
The concept of shared autoimmunity within individuals and families, sometimes referred to as the ‘‘kaleidoscope of autoimmunity,’’ has gained acceptance [19]. Co-existence of RA and psoriasis, multiple sclerosis, ulcerative colitis, lupus, type 1 diabetes mellitus and especially autoimmune thyroid disease (AITD) have been reported. Somers et al. conducted a series of population-based cohort studies, utilizing the United Kingdom General Practice Research Database, to assess intraindividual risks of coexistence of rheumatoid arthritis (RA), autoimmune thyroiditis (AIT), multiple sclerosis (MS), and insulin-dependent diabetes mellitus (IDDM) during 1990–1999. This study demonstrates coexistence of RA and AIT at higher than other diseases [19]. Conversely, Boelaert et al. found rheumatoid arthritis was the most common coexisting autoimmune disorder in Graves’ disease 3,15% and Hashimoto’s thyroiditis 4,24% to compare with many other autoimmune diseases such as type 1 diabetes, Addison ‘s disease, pernicious anemia, systemic lupus erythematosus, celiac disease, vitiligo, scleroderma, inflammatory bowel disease. [11] Although the exact pathogenic mechanisms of autoimmune disorders in AITD and RA are still not sufficiently defined, most of factors involved in the development of autoimmunity can be characterized as follows:
- genetic
- environmental—infectious agents, vaccines, smoking, drugs, stress factors, endocrine disruptors and others
Polymorphisms of HLA-DR B1, CTLA4, PTPN22, FCRL3 and IL2RA are common for both AITD and RA, whereas some are specific for one or for the other disease.[12].
RA affects 0,2% to 1,3% of the adult general population [2]. In Vietnam, RA prevalence is about 0,28% [15]. In Cho Ray hospital, RA accounts for 15% patients visit at the Rheumatology Department. The disease is three times more frequent in women than men. The prevalence rises with age and is highest in women older than 65 years[9].
The prevalence of thyroid dysfunction in the general population depends on various factors such as age, sex, geographical factors, and iodine intake. The number varies from 6% to 20% [20]. Sriphrapradang et al. . in 2009 estimated the prevalence of thyroid disorders in Thai population to be around 7,34% [3].
For several decades an increased occurrence of thyroid disorders in patients suffering from RA has been documented. A hormonal dysfunction and/or AITD are present in 8% to 35% patients with RA [8],[13],[18]. There are some impacts of thyroid dysfunction on RA patients that was shown in many researches. For example, Elatta et al. . found in his study that higher levels of TSH are associated with higher grades of RA disease activity (ESR, DAS28, and dose of methotrexate)[5].
Moreover, Ratermanet al. . investigated the risk of cardiovascular diseases in RA patients with hypothyroid abnormalities, the results showed in female RA patients, clinical hypothyroidism was associated with a fourfold higher risk of cardiovascular diseases in comparison with euthyroid female RA patients independently of the traditional risk factors [6]. Unrecognized and untreated thyroid dysfunction can lead to life-threatening clinical states, such as myxedema coma and thyroid storm. Unfortunately, because of asymptomatic course of AITD in the initial phase diagnosis might be difficult to be established[14].
Besides, AITD manifestations may resemble those presented by RA, and some symptoms are exacerbated when both diseases co-occur[10]. From all the reasons above, some authors has suggested to screen RA patients for thyroid diseases [1],[6],[11],[14],[18].
Although AITD and RA share common physiopathological mechanism, the connection between AITD and RA is a topic with no definite results so far. In Viet Nam, this association has not been explored. As one of the busiest center for Rheumatology in Hochiminh city, we would like to unravel this association in order to establish a solid base for future research in this area. The purpose of the study are:
- To determine the prevalence of thyroid dysfunction within RA patients.
- To describe characteristics of RA patients have thyroid dysfunction (any deviations on RA course due to thyroid diseases presence)
II. PATIENTS AND METHODS
Patients: the current study was conducted at Cho Ray Hospital. Patients attending the Rheumatology Department from Jan 2016 to June 2016 (outpatient and inpatient) diagnosed with rheumatoid arthritis where enrolled in the study. 113 patients were diagnosed with RA basis of ACR criteria 1987 and ACR/EULAR 2010 ( 06 males and 107 females).
In all the patients the following examinations were performed:
- Clinical examination: medical history taking (including history regarding thyroid diseases,RA and its familial occurrence), physical examination with internal examination and musculoskeletal system assessment
28 joints assessment according to EULAR criteria. Activity of RA was evaluated using the Disease Activity Score (DAS 28) with 4 parameters.
- Evaluation common thyroid symptoms include weight gain, inability to tolerate the cold, constipation, weight loss, palpitation, diarrhea, hair loss, irregular menstrual, decrease libido (all the symptoms appear 4 weeks prior to this investigation)
- Evaluation of goiter and its grade according to World Health Organization.
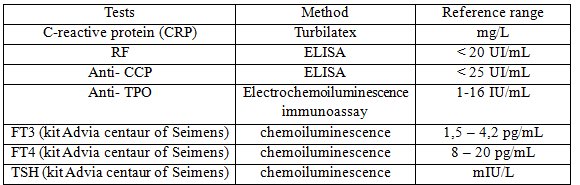
- Laboratory tests were performed at the Department of Biochemistry of Cho Ray Hospital, which already get the ISO 15189:2012, VILAS Med 045.
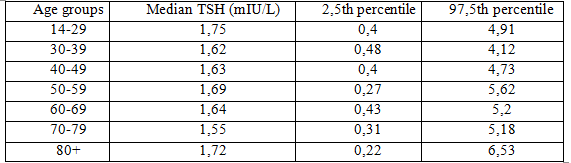
 TSH: 2,5th percentile ≤ TSH ≤ 97,5th percentile [3]
TSH: 2,5th percentile ≤ TSH ≤ 97,5th percentile [3]
- Thyroid dysfunction:
- Normal: normal TSH
- Subclinical hypothyroidism: high TSH but normal FT3,FT4
- Overt hypothyroidism: high TSH and low FT3,FT4
- Subclinical hyperthyroidism: low TSH but normal FT3,FT4
- Overt hyperthyroidism: low TSH and high FT3,FT4
- ATD diagnosis was made based on the presence of antithyroid antibodies (presence of anti-TPO antibodies was a prerequisite for the diagnosis) in patients with concomitant thyroid dysfunction and/or goiter.
Statistical analysis: Data analysis was done using Statistical Package for Social Sciences (SPSS software version-20). Means and standard deviations were calculated for quantitative data and proportions for categorical variables. To establish the relationship between categorical variables, Chi square testing was used and Independent t-test was used between continues variables with normal distribution assumed. Furthermore, regardless of categories, Pearson correlation Test was used to check the dependency of two continues variables. A P-value <0.05 is considered in rejecting null hypothesis.
III. RESULTS
1.Characteristics of RA patients
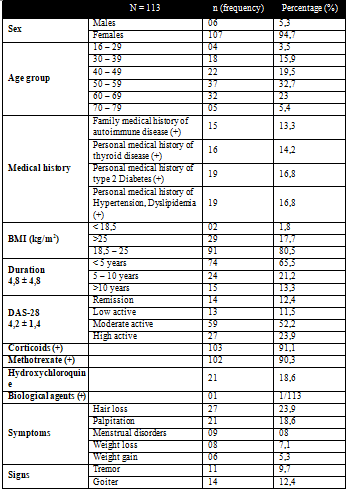
 2. Prevalence of thyroid diseases in RA
2. Prevalence of thyroid diseases in RA
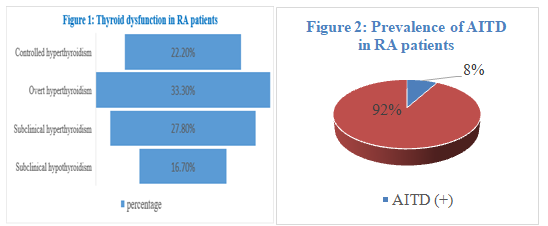
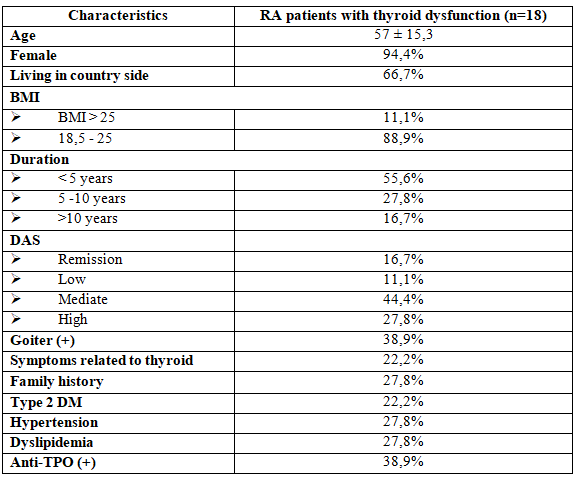
We found that the prevalence of thyroid dysfunction was 15,9% (18/113). The most common thyroid dysfunction observed was hyperthyroidism (83,3%). There was only 3 cases of subclinical hypothyroidism (16,7%) (figure 1). The presence of Anti-TPO was 23,9%. And the prevalence of AITD was 8%. (figure 2)
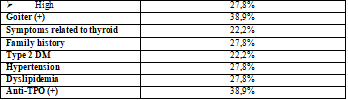
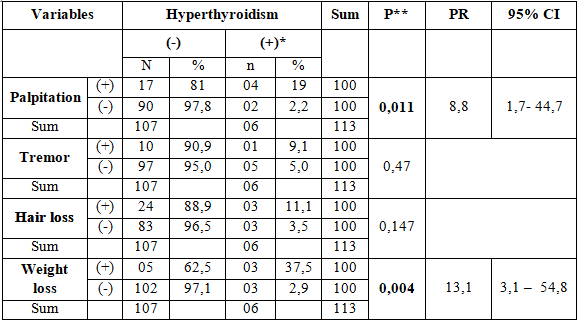
3. Characteristics of RA patients with thyroid dysfunction
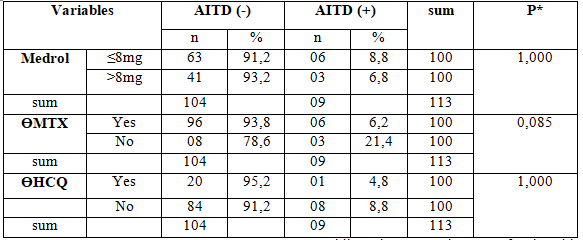
 Comparison between demographic and laboratory data of rheumatoid arthritis patients and RA patients with thyroid dysfunction doesn’t show any significant differences. This may due to the 18 abnormal cases have 5 sub-groups, and most of cases are subclinical. Therefore, we did the only bivariate analysis of the overt hyperthyroidism group (6 cases),we found the significance differences among palpitation and weight loss, age and use of medrol treatment
Comparison between demographic and laboratory data of rheumatoid arthritis patients and RA patients with thyroid dysfunction doesn’t show any significant differences. This may due to the 18 abnormal cases have 5 sub-groups, and most of cases are subclinical. Therefore, we did the only bivariate analysis of the overt hyperthyroidism group (6 cases),we found the significance differences among palpitation and weight loss, age and use of medrol treatment
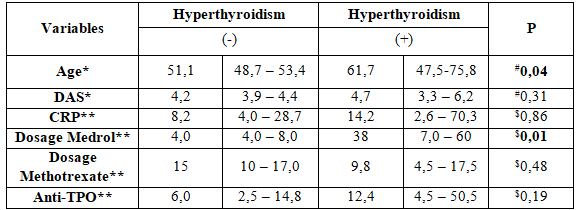 Note: Controlled hyperthyroidism and Subclinical hyperthyroidism were not included *mean (standard deviation), ** median (25%, 75%) #T student test$Mann-Whitney test.
Note: Controlled hyperthyroidism and Subclinical hyperthyroidism were not included *mean (standard deviation), ** median (25%, 75%) #T student test$Mann-Whitney test.
In bivariate analysis, significance difference among palpitation and weight loss, age and use of medrol treatment.
IV. DISCUSSION
While the prevalence of thyroid dysfunction in Thai general population was reported about 7,34%, the prevalence of thyroid dysfunction among Vietnamese patients with RA in this study is 15,9%. This is the summary of some recent observations about this issue:
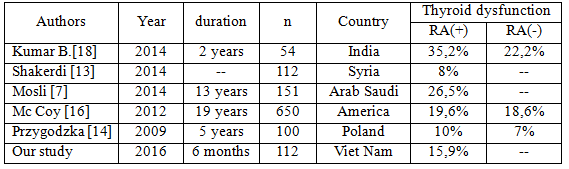 Significant differences in the prevalence of thyroid dysfunction in RA patients between studies may be explained by several factors. First of all, there is no universally consensus for the upper limit of normal TSH values, thus authors and clinicians worldwide accept different normal range. The second explanation for the spread frequency is due to iodine intake. Third, the time for taking samples also greatly affected the outcome.
Significant differences in the prevalence of thyroid dysfunction in RA patients between studies may be explained by several factors. First of all, there is no universally consensus for the upper limit of normal TSH values, thus authors and clinicians worldwide accept different normal range. The second explanation for the spread frequency is due to iodine intake. Third, the time for taking samples also greatly affected the outcome.
The most common thyroid dysfunction observed in this study was hyperthyroidism while in other studies hypothyroidism and subclinical hypothyroidism were the most common alterations among RA patients. The result was different to others but it is due to the race characteristic of thyroid disorders in Asian, as Weigle and MacLeod reported in their report about the variation of thyroid diseases in by ethnicity[4],[21].
The prevalence of positive Anti-TPO in our study was 23,9% , but the AITD prevalence was only 8%. This table below compares results between countries:
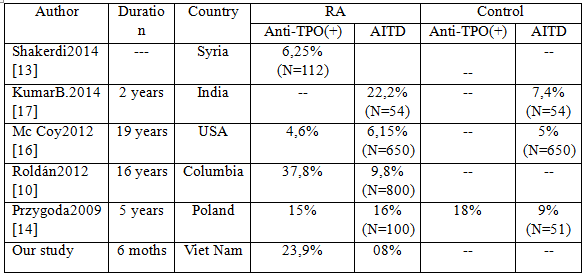 This high prevalence variability may be explain by certain factors. Firstly, there are difficulties on diagnosing AITD because it relies on the fact that there must be a positive of anti-thyroid a priority. Different methods for measuring Anti-TPO and the normal upper limit for Anti-TPO has not had the consensus. In our study, goiter was determined by clinical assessment instead of using thyroid ultrasound routinely in our study. This could lead to missing early changes in the structure of thyroid gland.
This high prevalence variability may be explain by certain factors. Firstly, there are difficulties on diagnosing AITD because it relies on the fact that there must be a positive of anti-thyroid a priority. Different methods for measuring Anti-TPO and the normal upper limit for Anti-TPO has not had the consensus. In our study, goiter was determined by clinical assessment instead of using thyroid ultrasound routinely in our study. This could lead to missing early changes in the structure of thyroid gland.
V. CONCLUSSION
Thyroid dysfunction was more frequent in RA patients. Subclinical hyperthyroidism and overt hyperthyroidism were the most common alterations among RA patients. Of note, thyroid dysfunction was clinically silent during the study as only 22,2% RA patients with concomitant thyroid disorders have symptoms related to thyroid diseases. It is clinically important to screen thyroid function for RA patients: over 40 years old, orwith weight loss, palpitation, goiter, high DAS-28 score, orusing high dose of glucocorticoids.
REFFERENCE
- Biondi B, Fazio, SCarella C, et al. (1993), “Cardiac effects of long term thyrotropin-suppressive therapy with levothyroxine. “, J Clin Endocrinol Metab, 77334-77338.
- CDC, Centers for Disease Control and Prevention. Rheumatoid Arthritis. 2016; Available from: https://www.cdc.gov/arthritis/basics/rheumatoid.htm.
- Chutintorn Sriphrapradang, Supatra Pavarangkoon, Wallaya Jongjaroenprasert, et al. (2014), “Reference ranges of serum TSH, FT4 and thyroid autoantibodies in the Thai population: the national health examination survey”, Clinical Endocrinology (2014) 80, 751–756, 80, 751-756.
- Donald S. A. McLeod, Patrizio Caturegli, David S. Cooper, et al. (2014), “Variation in Rates of Autoimmune Thyroid Disease by Race/Ethnicity in US Military Personnel”, JAMA. , 311 (15), 1563-1565.
- Enas A. Elattar, Takwa B. Younes, Sameh A. Mobasher (2014), “Hypothyroidism in patients with rheumatoid arthritis and its relation to disease activity”, Egyptian Rheumatology and Rehabilitation, 41 (2), 58-65.
- H.G.Raterman, et al (2008), “Rheumatoid arthritis is associated with a high prevalence hypothyroidism which amplifies its cardiovascular risk”, Annals of Rheumatic Diseases, 67 (2), 229-332.
- Hala H. Mosli, Suzan M. Attar (2014), “Prevalence and Patterns of Thyroid Dysfunction in Patients with Rheumatoid Arthritis”, The Open Endocrinology Journal, 7, 1-5.
- Hala H. Mosli, Attar and Suzan M. (2014), “Prevalence and Patterns of Thyroid Dysfunction in Patients withRheumatoid Arthritis”, The Open Endocrinology Journal, 7, 1-5.
- Howard R Smith, Brown. Adam. Rheumatoid Arthritis. 2016; Available from:http://emedicine.medscape.com/article/331715-overview.
- Jorge Cardenas Roldán, Jenny Amaya-Amaya, Juan Castellanos-de la Hoz, et al. (2012), “Autoimmune Thyroid Disease in Rheumatoid Arthritis: A Global Perspective”, Arthritis, 2012
- Kristien Boelaert, Paul R. Newby, al et (2010), “Prevalence and Relative Risk of Other Autoimmune Diseases in Subjects with Autoimmune Thyroid Disease”, The American journal of medicine 123 (2), 1-9.
- Lazúrová I, Jochmanová I, Benhatchi K, et al. (2014), “Autoimmune thyroid disease and rheumatoid arthritis: relationship and the role of genetics.”, Immunol Res. , 60 (2-3), 193-200.
- Loai Shakerdi, Wadah Haj Naema, & Saied Hamdon (2014), “Prevalence of thyroid dysfunction in patients with rheumatoid arthritis”, Endocrine Abstracts, 34, 386.
- Przygodzka M, Filipowicz-Sosnowska A. (2009), “Prevalence of thyroid diseases and antithyroid antibodies in women with rheumatoid arthritis”, Pol Arch Med Wewn., 119 (1-2), 39-43.
- Rose Wong, Aileen M.Davis, Elizabethe Badley, Prevalence of Arthritis and Rheumatic diseases around the world: a growing burden and implications for health care needs, 2010, Arthritis Community Research and Envaluation Unit: Toronto.
- Sara McCoy M.D., Cynthia S. Crowson M.S., Sherine E. Gabriel M.D., M.Sc., et al. (2013), “Hypothyroidism as a risk factor for development of cardiovascular disease in patients with rheumatoid arthritis”, J Rheumatol.
- Siddhartha Kumar B, Sivaram Naik G, Mohan A Prabath Kumar D, et al. (2014;), “Prevalence of thyroid disorders and metabolic syndrome in adult patients with rheumatoid arthritis”, J Clin Sci Res, 3, 97-105.
- Siddhartha Kumar B, Sivaram Naik G, Alladi Mohan, et al. (2014), “Prevalence of thyroid disorders and metabolic syndrome in adulsts patients with Rheumatoid arthritis”.
- Somers EC, Thomas SL, Smeeth L, et al. (2006), “Autoimmune diseases co-occurring within individuals and within families: a systematic review”, Epidemiology. , 17 (2), 202-217.
- Vanderpump Mark P. J. (2011), “The epidemiology of thyroid disease”, Oxford Journals Medicine & Health, 99 (1), 39-51.
- Weigle DS, Hooton TM, Toivola B, et al. (1996), “Frequency of thyroid disease among Southeast Asian primary care patients”, J Clin Pharmacy and Therapeutic, 21 (1).
 Hội Nội Tiết – Đái Tháo Đường Miền Trung Việt Nam Hội Nội Tiết – Đái Tháo Đường Miền Trung Việt Nam
Hội Nội Tiết – Đái Tháo Đường Miền Trung Việt Nam Hội Nội Tiết – Đái Tháo Đường Miền Trung Việt Nam