HEART FAILURE IN TYPE 2 DIABETIC PATIENTS
WITH SILENT ISCHEMIC HEART DISEASE
Nguyen Do Thanh Dat, Nguyen Hai Thuy
Hue Unviversity of Medecine and Pharmacy
ABSTRACT
Objectives: Type 2 Diabetes mellitus is a disease that is strongly associated with cardiovascular complications, including ischemic heart disease (IHD), the aims of this study is to determine the prevalence of HF in type 2 diabetic patients with silent ischemic heart disease (SIHD) and the risk factors of HF in those patients. Patients and Methods: A total of 51 type 2 diabetic patients who were diagnosed SIHD based on ECG and/or echocardiography and/or coronary angiogram and without chest pain, evaluated cardiovascular risk factors. Results: The prevalence of heart failure in T2DM patients with SIHD was 23.5% (12/51 patients), 10/12 patients had systolic heart failure and 6/12 patients with heart failure had combinations of both LV diastolic and systolic dysfunction. LV diastolic and systolic dysfunction had rigorously significant relation with heart failure. Previous MI was the major cause of LV systolic dysfunction and LV wall motion abnormalities. Male and smoking may be seen as the risk factors of MI in T2DM patients with SIHD. Conventional lipid parameters, lipoprotein ratios were not predictor of MI in T2DM patients with SIHD. However, not all patients with previous MI develop LV systolic dysfunction. So a role of timely diagnosis, early percutaneous coronary intervention are the most important for the patients with acute MI. Left ventricular diastolic dysfunction, an early sign of diabetic cardiomyopathy, and cardiac autonomic neuropathy contributed to higher prevalence and severer of congestive HF in T2DM patients with SIHD.
Main correspondence: Nguyen Do Thanh Dat
Submission date: 1st August 2018
Revised date: 18th August 2018
Acceptance date: 31th August 2018
1.INTRODUCTION
Diabetes mellitus is a disease that is strongly associated with cardiovascular complications, including ischemic heart disease (IHD), peripheral arterial disease, stroke, cardiomyopathy, and congestive heart failure (HF). Abundant evidence shows that there is an increased risk of cardiovascular disease mortality and morbidity in patients with type 2 diabetes mellitus, particularly IHD.
The IHD in diabetic patients is characterized by accelerated atherosclerosis occurring at an earlier age than the nondiabetic population. It is more extensive, diffuse inmultiple arterial territories, and involves more distal and smallercoronary arteries [10]. Perhaps the overriding risk factor for IHD in insulin resistance and T2DM is the associated dyslipidemia [13]. The autonomic nervous system, which controls a widerange of bodily functions, can be damaged in diabetes with a variety of manifestations, most commonly, cardiovascular, urogenital, gastrointestinal, thermoregulatory, and sudomotor function [9].
Cardiac autonomic neuropathy manifestsinitially as an increase in heart rate secondary to vagal denervation, followed by a decrease due to sympathetic denervation; fnally, a fxed heart rate, which respondsonly minimally to physiologic stimuli, supervenes, bearing similarities to the transplanted heart and suggesting almost complete denervation [9]. T2DM increases the risk of silent ischemic heart disease (SIHD), result in delayed diagnosis, or undiagnosed episodes of silent ischemia.
So the patientsare not received effective and timely medical managements, and able to have serious consequences, including adverse medical outcomes, even death. Particularly, the prevalence of unrecognized or silent MI appears to be higher in patients with diabetes mellitus compared with nondiabetics, ranging from 2% to 7% [11].
Left ventricular (LV) systolic dysfunction only established when damage or death of significant part of myocardium occurs as a result of acute myocardial infarction [1]. The more myocardium lose, the more LV systolic function is reduced [1].
Ischemic cardiomyopathy is determined when ejection fraction (EF) < 50% (is considered as reduced EF). Although ICH is recognized a double of the incidence and severer in patients with DM, it is not common have extensive infarction when evaluated by the release of creatine kinase, radionuclide ventriculography or echocardiography [1].
To improving prognosis and outcomes in diabetic patients with SIHD, an early diagnosis and appropriate management are the most important. So this study was designed with two purposes.
Objectives
To determine the prevalence of HF in type 2 diabetic patients with SIHD and the risk factors of HF in those patients.
Patients and Methods
1.1. Study population
A total of 51 type 2 diabetic patients who was diagnosed SIHD based on ECG and/or echocardiography and/or coronary angiogram and without chest pain.
Exclusion criteria are patients disagree to participate in the study, type 1 and other types diabetes mellitus, and be acquired other cardiovascular diseases, including valvular heart diseases (mitral valve stenosis, mitral valve regurgitation, aortic valve stenosis, aortic valve regurgitation,…), cardiomyopathy (dilated cardiomyopathy, hypertrophic cardiomyopathy, restrictive cardiomyopathy, arrhythmogenic right ventricular dysplasia,…), cardiopulmonary diseases (pulmonary hypertension, pulmonary embolism, sleep apnea,…), or other critical illnesses such as: septic shock, stroke, kidney failure, acute respiratory distress syndrome,…
1.2. Diagnostic criteria
IHD is diagnosed when has one or more criteria, including ECG has horizontal or downsloping ST segment depression of at least 1 mm and/or echocardiography has the induction of a regional wall motion abnormality and/or coronary angiogram has significant coronary stenosis (50% or greater lumen stenosis of the left main coronary artery or 70% or greater lumen stenosis of the left anterior descending or the left circumflex or the right coronary artery).
Ischemic cardiomyopathy has been defined when patients have IHD and LV systolic dysfunction (EF<50%). Using the Framingham criteria for diagnosis of HF. Hypertension was defined as the measurement of systolic/diastolic blood pressure higher than 140/90 mmHg. T2DM was diagnosed based on ADA 2017 guideline and/or had been previously diagnosed T2DM and was using anti-hyperglycemic medications or insulin.
1.3. Statistical analysis
Continuous variables are presented as the mean ± standard deviation (SD) and were compared using an independent samples ttest. Categorical variables are expressed as frequencies and percentages and were compared using a Chi-square test.
The correlation between variables was calculated by a Pearson correlation analysis. All statistical analyses were performed with Statistical Package for the Social Sciences 22.0 (IBM SPSS Statistics 22.0). A value of P<.05 in a 2-sided test was considered significant.
2. RESULTS
Table 1. Anthropometric, biochemical parameters for T2DM patients with SIHD
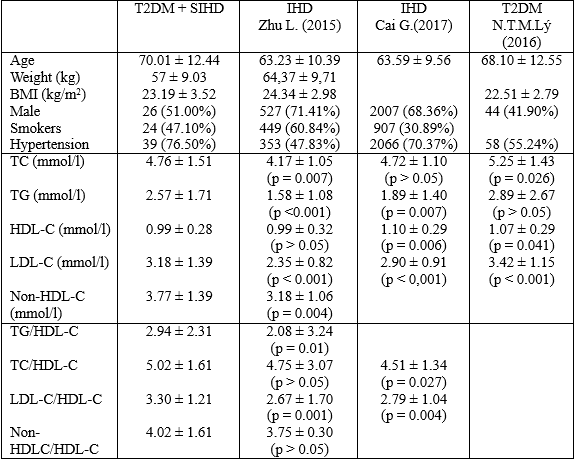
Table 2. Classification of Heart Failure

* Heart failure with preserved (HFpEF), mid-range (HFmrEF), and reduced (HFrEF) ejection fraction
Table 3. Left ventricular systolic dysfunction and diastolic dysfunction for patients
with and without heart failure
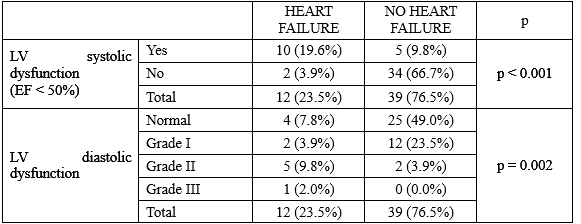
Table 4. Left ventricular systolic dysfunction and diastolic dysfunction for heart failure
in T2DM patients with SIHD
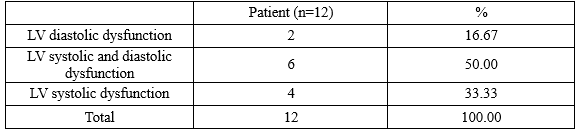
Table 5. The risk factors for LV systolic dysfunction in T2DM patients with SIHD
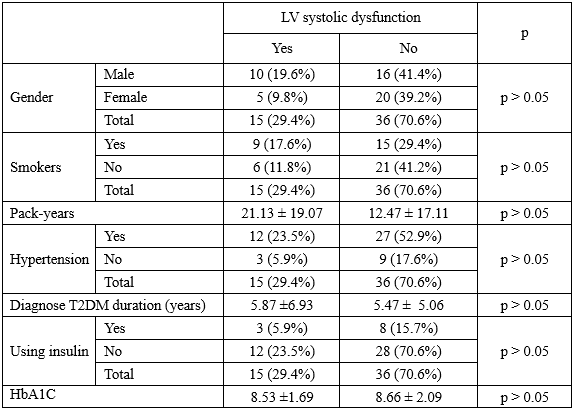
Table 6. Electrocardiogram and echocardiography for LV systolic dysfunction
in T2DM patients with SIHD
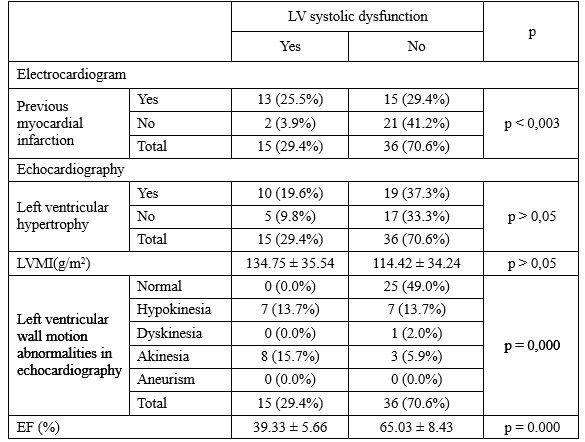
Table 7. The risk factors for previous myocardial infarction in T2DM patients with SIHD
Table 8. Lipid parameters, and lipoprotein ratios for previous myocardial infarction
in T2DM patients with SIHD
 Table 9. The risk factors and echocardiography for left ventricular diastolic dysfunction
Table 9. The risk factors and echocardiography for left ventricular diastolic dysfunction
in T2DM patients with SIHD
The importance of the atherogenic lipoprotein particles, including LDL, VLDL, IDL, and Lp(a) in the development of atherosclerosis, particularly IHD (IHD), and anti-atherogenic role of HDL in preventing atherosclerosis diseases have long been recognized in numerous studies. LDL cholesterol remains the primary target of therapy for the prevention of IHD. In recent studies have illustrated that the lipoprotein ratios and non-HDL-C are good predictors of vascular diseases; which have greater power than isolated lipoprotein parameters used independently, particularly LDL-C and similar power to that of the apoB/apoA-I ratio.
In a hospital – based case-control study consisting of 738 IHD patients and 157 control subjects was conducted in a Chinese Han population, Zhu L. (2015) showed that the IHD group had significantly higher of non-HDL-C, TG/HDL-C, TC/HDL-C, LDL-C/HDL-C, non-HDL-C/HDL-C than the control group (p<0.05 for all).
However, there were no significant differences in TG, TC, LDL-C, HDL-C, LDL-C, apoAI, and apoB100 between the two groups [8]. In a similar study, Cai G. (2016) indicated that the value of nontraditional lipid profiles, including non-HDL-C, TC/HDL-C, LDL-C/HDL-C, and atherogenic index of plasma, were all significant higher in the IHD group than in the control group, and might be a strong and independent predictor for IHD in the Chinese Han population [3].
In our study, there were no values of lipid parameters, lipoprotein ratios, and non-HDL-C in T2DM patients with SIHD which were lower than the IHD group in Zhu’s study (2015).
Specially, the values of TC, TG, LDL-C, non-HDL-C, TG/HDL-C, and LDL-C/HDL-C were all significantly higher in T2DM patients with SIHD than in the IHD group.
Compared with IHD group in Cai’s study (2016), the T2DM patients with SIHD had significantly higher values of TG, LDH-C, TC/HDL-C, LDL-C/HDL-C, significantly lower value of HDL-C. And there was no significant difference in TC between two groups. However, compared with T2DM patients in Nguyen Hai Thuy, Nguyen Thi Minh Ly study (2016), the concentration of TC, LDL-C, HDL-C were all significant lower. To explain this situation whether T2DM patients with SIHD were received more aggressive pharmacologic treatment of dyslipidemia, particularly statin therapy, than the T2DM patients alone.
A total of 51 T2DM patients with SIHD took part in this study, there were 12 patients (23.5%) diagnosed with heart failure (HF) based on Framingham diagnostic criteria and European Society of Cardiology criteria. About classification of HF, the number of patients had HF with preserved (HFpEF), mid-range (HFmrEF), and reduced (HFrEF) ejection fraction were 2 (3.9%), 5 (9.8%), 5 (9.8%), respectively. There were 10/12 patients (83.33%) diagnosed with HF had reduced left ventricular (LV) systolic dysfunction and only had 2 patients with heart failure had isolated LV diastolic dysfunction. So the most common cause of heart failure was LV systolic dysfunction or reduced LV ejection fraction.
Table 3.3 demonstrated that there were the statistically significant relation between ischemic cardiomyopathy (LV systolic dysfunction), LV diastolic dysfunction and HF with p < 0.001, 0.002, respectively. So it meant that the T2DM patients with SIHD diagnosed with heart failure often had combinations of both LV systolic and diastolic dysfunction, with 6/12 patients (50.0%).
This is a reason why diabetic patients have higher prevalence of severe HF and have a significant higher risk of mortality and morbidity at hospital due to HF than non-diabetic patients.
Talking about LV systolic dysfunction in T2DM patients with SIHD, in our study, there were 15 patients (29.4%) had LV systolic dysfunction with EF < 50%. Male, smokers, hypertension patientswere more likely to have LV systolic dysfunction with 10/15 patients (66.7%), 9/15 patients (60.0%), and 12/15 patients (80.0%).
However, there were no statistically significant differences between males and females; between smokers and non-smokers; between hypertension and normotension were observed in the risk factors for LV systolic dysfunction in the T2DM patients with SIHD (p > 0.05). The known T2DM duration were no differences between two groups with and without LV systolic dysfunction, 5.87 ± 6.93, and 5.47 ± 5.06 years (p > 0.05), respectively.
However, Rae DC indicated that myocardial damage in patients with diabetes affected diastolic function before systolic function, diastolic dysfunction was seen in diabetic patients approximately 8 years after onset of diabetes and systolic dysfunction approximately 18 years [1].
So it meant that our patients was no early diagnosed at the earliest stage of T2DM. Therefore, the patients were not controlled their blood glucose level to prevent cardiovascular complications.
Additionally, HbA1C level was high and no significant difference between two group with and without LV systolic dysfunction, 8.53 ± 1.69, and 8.66 ± 2.09% (p > 0.05), respectively.
Diabetic men in the Multiple Risk Factor Intervention Trial (MRFIT) had an absolute risk of IHD death more than three times higher than that of the nondiabetic cohort, even after adjustment for established risk factors.
Seminal work from Finland showed that patients with T2DM without a previous myocardial infarction (MI) have a risk of MI over 7 years as high as that of nondiabetic patients with a history of MI [13]. In our study, 13/15 T2DM patients (86.7%) with LV systolic dysfunction detected by a medical history and ECG had previous MI.
And there were rigorously significant relation between previous MI and LV systolic infarction with p < 0.003.
Compared with the group had normal LV systolic function, group had LV systolic dysfunction had significant higher prevalence of LV wall motion abnormalities in echocardiography with 15/15 patients, and 11/36 patients, respectively. And LV wall motion abnormalities had statistically significant relate with LV systolic dysfunction (p = 0.000). LV ejection fraction (EF) was significantly lower in the patients with LV systolic dysfunction than the patient had normal LV systolic function, with 39.33 ± 5.66, 65.03 ± 8.43% (p = 0.000). Overall, previous MI caused a loss of contractile fibers and LV fibrosis which leaded to LV wall motion abnormalities, reduced EF, and finally result in systolic HF. LV mass index was no different between two groups, and LV hypertrophy did not relate with LV systolic dysfunction.
What is the risk factor of MI in T2DM patients with SIHD? Table 3.7 illustrated that males were at higher risk of MI than females, and male significantly related to MI with p = 0.036. In general, T2DM patients with previous MI smoked more than ones no previous MI, with 20.07 ± 19.08, and 8.87 ± 14.64 pack-years, respectively.
There were significant differences between two group with and without previous MI about the known T2DM duration, HbA1C, traditional lipid parameters, non-HDL-C, and lipoprotein ratios. Hypertension did not relate to previous MI in T2DM patients with SIHD. Finally, male and smoking able to be seen as the risk factors of MI in T2DM patients with SIHD.
Diabetes is associated with a fourfold increase in the risk of congestive HF, even after adjustment for other cardiovascular risk factors such as age, BP, cholesterol level, obesity, and history of IHD [13]. Patients with diabetes, who commonly present with either HFpEF, HFmrEF, or HFrEF, experience higher rates of congestive HF than donondiabetic patients after an acute MI, regardless of thesize of the infarct zone [13]. These fndings suggest that diabetes it self causes deleterious effects on the myocardium, leading to poorer outcomes [13].
Besides LV systolic dysfunction due to MI, diabetic cardiomyopathy also contributed to higher prevalence and severer of congestive HF in diabetic patients than non-diabetic patients.
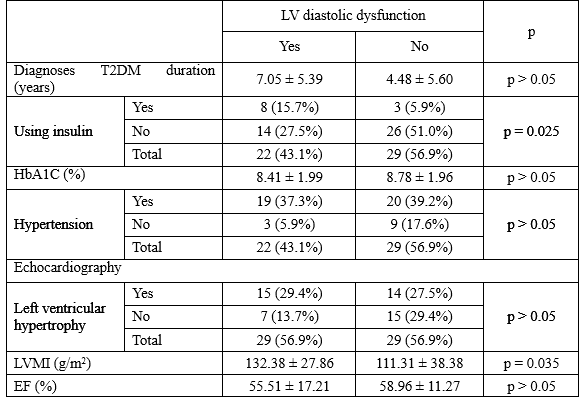
Diabetic cardiomyopathy affects the myocardium in diabetic patients, result in a wide range of structural abnormalities, initially characterized by myocardial fibrosis, dysfunctional remodeling, and associated dysfunction, later by systolic dysfunction, and eventually by congestive HF. Diastolic dysfunction has been recognized as an early sign of this diabetic myocardium disease. Additionally, the T2DM patients with SIHD always combine with cardiac autonomic neuropathy, which is known to contribute to both systolic and diastolic dysfunction. Diastolic dysfunction often exist after 8 years from the onset of diabetes mellitus [1]. In our study, there were 22/51 patients (43.1%) had LV diastolic dysfunction.
The patients had LV diastolic dysfunction had the diagnoses T2DM duration with 7.05 ± 5.39 years, compared with 4.48 ± 5.60 years of the patients with normal LV diastolic function, but the difference is no statistically significant. The patients using insulin were more likely had LV diastolic dysfunction and this relationship was statistically significant with p = 0.025. After 10 to 15 years from the onset of T2DM, almost patients with need insulin and that are enough time to exist LV diastolic dysfunction in T2DM patients. Although coexisting hypertension and diabetes may contribute to the appearance of diastolic dysfunction common in diabetic patients.
However, in our study, LV diastolic dysfunction were independent with hypertension and HbA1C. On echocardiography, the T2DM patients with diastolic dysfunction had significant higher of LM mass index with 132.38 ± 27.86, and 111.31 ± 38.38 g/m2, respectively. Finally, there were not significant difference about EF between two groups with and without LV diastolic function.
- CONCLUSIONS
The prevalence of heart failure in T2DM patients with SIHD was 23.5% (12/51 patients), 10/12 patients had systolic heart failure and 6/12 patients with heart failure had combinations of both LV diastolic and systolic dysfunction. LV diastolic and systolic dysfunction had rigorously significant relation with heart failure.
Previous MI was the major cause of LV systolic dysfunction and LV wall motion abnormalities. Male and smoking may be seen as the risk factors of MI in T2DM patients with SIHD. Conventional lipid parameters, lipoprotein ratios were not predictor of MI in T2DM patients with SIHD. However, not all patients with previous MI develop LV systolic dysfunction. So a role of timely diagnosis, early percutaneous coronary intervention are the most important for the patients with acute MI.
Left ventricular diastolic dysfunction, an early sign of diabetic cardiomyopathy, and cardiac autonomic neuropathy contributed to higher prevalence and severer of congestive HF in T2DM patients with SIHD.
REFERENCES
- Nguyễn Hải Thủy (2016), “Cập nhật chẩn đoán và điều trị bệnh cơ tim đái tháo đường năm 2016“, Nội tiết – Đái tháo đường. 21, pp. 62-83.
- Nguyễn Hải Thủy, Nguyễn Thị Minh Lý (2016), Nghiên cứu Bệnh tim Đái tháo đường ở bệnh nhân đái tháo đường typ 2, Thủy, ed, Trường Đại học Y Dược Huế.
- Cai G., Shi G., Xue S., Lu W. (2017), “The atherogenic index of plasma is a strong and independent predictor for coronary artery disease in the Chinese Han population“, Medicine (Baltimore). 96(37), pp. e8058.
- Kang Y. M., Kim Y. J., Park J. Y., Lee W. J., et al (2016), “Mortality and causes of death in a national sample of type 2 diabetic patients in Korea from 2002 to 2013“, Cardiovasc Diabetol. 15(1), pp. 131.
- Nguyen C. T., Pham N. M., Lee A. H., Binns C. W. (2015), “Prevalence of and Risk Factors for Type 2 Diabetes Mellitus in Vietnam: A Systematic Review“, Asia Pac J Public Health. 27(6), pp. 588-600.
- Nguyen T. K. (2015), “Diabetes in Vietnam“, Ann Glob Health. 81(6), pp. 870-3.
- Tseng C. H. (2004), “Mortality and causes of death in a national sample of diabetic patients in Taiwan“, Diabetes Care. 27(7), pp. 1605-9.
- Zhu L., Lu Z., Zhu L., Ouyang X. , et al (2015), “Lipoprotein ratios are better than conventional lipid parameters in predicting coronary heart disease in Chinese Han people“, Kardiol Pol. 73(10), pp. 931-8.
- Andrew J.M. Boulton ,Rayaz A. Malik (2016), “Diabetes Mellitus: Neuropathy “Endocrinology : adult & pediatric – Seventh Edition, Elsevier, Philadelphia, pp. 920-934.
- Barak Zafrir, Jorge Plutzky (2015), “Atherogenesis, coronary heart disease and insulin resistance syndrome in diabetes” International Textbook of Diabetes Mellitus – Fourth Edition, John Wiley & Sons Ltd, Oxford, UK,pp. 1031-1046.
- Benjamin M. Scirica (2013), “Prevalence, Incidence, and Implications of Silent Myocardial Infarctions in Patients With Diabetes Mellitus“, Circulation. 127, pp. 965-967.
- Jozo Boras, et al (2010), “Silent Ischemia and Diabetes Mellitus“, Diabetologia Croatica. 39(2), pp. 57-65.
- Kenneth S. Polonsky, Charles F. Burant (2016), “Type 2 Diabetes Mellitus” Williams textbook of endocrinology – Thirteenth edition, Elsevier, Philadelphia, pp. 1386-1450.
- Peter F. Cohn, Kim M. Fox, Caroline Daly (2003), “Silent Myocardial Ischemia“, Circulation. 108(9), pp. 1263-1277.
15.Boussageon R, Bejan-Angoulvant T, Saadatian-Elahi M, et al.Effect of intensive glucose lowering treatment on all cause mortality, cardiovascular death, and microvascular events in type 2 diabetes: meta-analysis of randomised controlled trials. BMJ 2011;343:d4169.
- ACCORD Study Group, Gerstein HC, Miller ME, Genuth S, et al. Long-term effects of intensive glucose lowering on cardiovascular outcomes. N Engl J Med 2011;364:818–28.
- Ukena C, Dobre D, Mahfoud F, et al. Hypo- and hyperglycemia predict outcome in patients with left ventricular dysfunction after acute myocardial infarction: data from EPHESUS. J Card Fail 2012;18: 439–45.
- MacDonald MR, Eurich DT, Majumdar SR, et al. Treatment of type 2 diabetes and outcomes in patients with heart failure: a nested case-control study from the U.K. General Practice Research Database. Diabetes Care 2010;33:1213–8.
- Boussageon R, Supper I, Bejan-Angoulvant T, et al. Reappraisal of metformin efficacy in the treatment of type 2 diabetes: a meta-analysis of randomised controlled trials. PLoS Med 2012;9:;
- Monami M, Dicembrini I, Mannucci E. Dipeptidyl peptidase-4 inhibitors and heart failure: a meta-analysis of randomized clinical trials. Nutr Metab Cardiovasc Dis 2014;24:689–97.
- Savarese G, Perrone-Filardi P, D’Amore C, et al. Cardiovascular effects of dipeptidyl peptidase-4 inhibitors in diabetic patients: a meta-analysis. Int J Cardiol 2015;181:239–44.
- Green JB, Bethel MA, Armstrong PW, et al. Effect of Sitagliptin on Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med 2015;373(3):232–42.
 Hội Nội Tiết – Đái Tháo Đường Miền Trung Việt Nam Hội Nội Tiết – Đái Tháo Đường Miền Trung Việt Nam
Hội Nội Tiết – Đái Tháo Đường Miền Trung Việt Nam Hội Nội Tiết – Đái Tháo Đường Miền Trung Việt Nam